Abstract
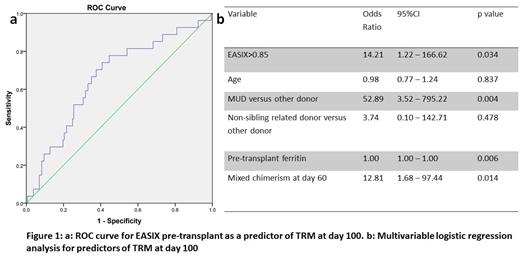
Allogeneic stem cell transplantation (SCT) is a curative treatment for beta-thalassemia major. The Pesaro risk stratification classifies patients with thalassemia into three groups (class I, II, and III) to predict the transplant-related mortality (TRM) and graft rejection after myeloablative conditioning with busulfan and cyclophosphamide. However, in developing countries, where inadequate chelation therapy prior to transplant is common, most patients would fall under the class III category. There is heterogeneity in the clinical outcomes in this group. Hence, our center introduced a further refinement in this risk stratification, identifying a subset of "class III high risk" as those with age greater than or equal to 7 years and liver size > 5cm (Mathews V et al. BBMT 2007). Subsequently, we showed that the use of a conditioning regimen containing thiotepa, treosulfan, and fludarabine (TTF) along with a peripheral blood stem cell graft led to improved outcomes in this subset with a reduction in early TRM from 46% (with busulfan-cyclophosphamide) to 13% (Mathews V et al. PLoS One 2013). There is still a need to identify predictors of poor clinical outcomes to optimize further the clinical outcomes of these class III high-risk thalassemia patients. Endothelial activation and stress index (EASIX) is a simple biomarker calculated using lactate dehydrogenase, creatinine, and platelet counts. EASIX has been shown to be predictive of overall survival in various settings like GVHD following reduced-intensity transplants for malignancies in adults, veno-occlusive disease, and nonrelapse mortality following SCT (Luft T et al. Lancet Haem 2017; Jiang S et al. Haematologica 2021; Luft T et al. BMT 2020). Here, we evaluated the role of EASIX (measured before conditioning therapy) as a biomarker in predicting early TRM in patients with thalassemia major who have undergone SCT with a uniform conditioning regimen using TTF at our center. During the study period from January 2012 to December 2019, 281 patients with thalassemia major underwent SCT with a uniform TTF protocol at our center. The median age was nine years (range 1 to 25 years). One hundred and nine (38.8%) were females. As per Pesaro classification (with Vellore modification), three (1.1%) were class I, 34 (12.1%) were class II, 134 (47.7%) were class III low risk, and 110 (39.1%) were class III high risk. The stem cell donors were matched sibling (n=218, 77.6%), matched related non-sibling (n=23, 8.2%), or matched unrelated donors (n=40, 14.2%). The stem cell donor was HLA matched in all cases except 21 (7.5%), wherein there was a mismatch at one locus. Five (1.8%) had a bone marrow graft, while others had peripheral blood stem cell grafts. Thirty-eight (13.5%) patients had transplant-related mortality by day 100 (TRM100). The median follow-up of the cohort was 31 months (range 0 to 103 months). EASIX score pre-transplant was available for 184 (65.5%) patients. There was no difference in the rate of TRM100 in patients where EASIX was available compared to those where EASIX was not available (14.7% versus 11.3%, p 0.47). Also, there was no difference in overall mortality rate in patients where EASIX was available compared to those where EASIX was not available (21.2% versus 17.5%, p 0.53). Among patients with TRM100 vis-à-vis those who did not, the median EASIX score was significantly higher (1.09 versus 0.75, p 0.008). We then plotted a receiver operating characteristics (ROC) curve for predicting TRM100 using the EASIX score. The area under the curve was 0.661 (Figure 1a). A cut-off of 0.85 for the EASIX score had a 70.4% sensitivity and 62% specificity for predicting TRM100. The TRM100 for patients with EASIX above 0.85 was significantly higher than those with EASIX less than 0.85 (24.4% versus 7.5%, p 0.003). On multivariable logistic regression analysis, the factors independently predicting TRM100 were pre-transplant EASIX score, pre-transplant ferritin, unrelated donor source, and chimerism at day 60 (Figure 1b). In patients with thalassemia major undergoing SCT using a uniform TTF conditioning, EASIX score measured pre-transplant can identify patients at greater risk for Day100 TRM.
Mathews: Christian Medical College: Patents & Royalties: US 2020/0345770 A1 - Pub.Date Nov.5, 2020; AML: Other: Co-Inventor.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal